
Making stuff cool and keeping it that way has been a pretty essential part of human civilization for thousands of years, with only in the past few hundred years man-made methods having become available that remove the reliance on the whims of nature and lugging around massive blocks of ice. The most important cooling method is undoubtedly that of vapor-compression refrigeration, but this is hardly the only method to transfer thermal energy from one location to another.
For example, we recently covered an elastocaloric cooling project by a group of scientists that uses strips of NiTi metal. By flexing these they induce a cooling effect which when put in a number of stages serves to transfer a significant amount of thermal energy between both sides, much like a vapor-compression system but without the gases and compressor. Meanwhile the Seebeck effect is relatively well-known from Peltier thermocouple devices, and features heavily in portable refrigerators and kin where these solid-state devices can also transfer thermal energy.
Of course, along with how they function the major question with all of these cooling technologies is how efficient they are, as this determines when you’d want to even consider them for a specific application.
The Science Of Cold
Although as animals we have an intuitive understanding of what concepts like ‘cold’ and ‘hot’ are in the sense of comfort levels, on a fundamental level the related concept of temperature is about the kinetic energy of the particles in a system. Essentially, the more kinetic energy exists in the system, the higher the temperature of said system is, regardless of whether it’s a liquid, gas, solid or plasma. Hence a temperature of zero Kelvin is the complete absence of any such kinetic energy in the system, also known as the Third Law of thermodynamics:
As the temperature of a system approaches absolute zero, all processes cease and the entropy of the system approaches a minimum value.
When we talk about moving thermal energy from one location to another – as in refrigeration – this thus means transferring said energy from one system to another in some fashion, something which is covered by the First Law of thermodynamics:
In a process without transfer of matter, the change in internal energy,, of a thermodynamic system is equal to the energy gained as heat, , less the thermodynamic work,
, less the thermodynamic work, , done by the system on its surroundings.
, done by the system on its surroundings.
In the case of a hot water bottle or ice bag we are actively changing the energy balance of a system by transferring matter. This makes such transfers rather lossy, which is not a quality that is generally desirable in a refrigeration system. Thus we prefer a closed system in which the matter is ideally never lost, and thus all the energy transfer occurs via reversible processes.
Vapor-Compression
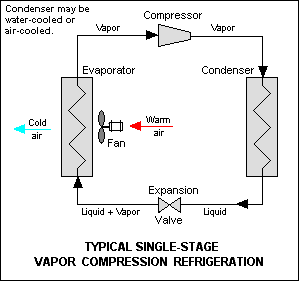
Single-stage vapor-compression refrigeration system components. (Credit: mbeychok, Wikimedia)
In vapor-compression refrigeration a liquid – the refrigerant – is circulated through the system, alternately changing state into a gas by absorbing thermal energy from the environment, before shedding this energy again while condensing back into a liquid.
A key component in this system is the compressor, which takes in the saturated vapor. This means that said vapor contains enough energy to effect the liquid-gaseous transition, but is still pretty close to the condensing point.
By compressing this vapor into a smaller volume its temperature increases since roughly the same amount of kinetic energy exists within the system. This superheated vapor then passes through the condenser, like the radiator found at the back of the average kitchen refrigerator. Here the superheated vapor condenses back into a liquid, with the higher temperature and pressure helping to make the condensing process more efficient. This is also why said refrigerator radiator can feel so warm to the touch.
The role of the expansion valve is effectively the opposite of the compressor: as the name suggests this is where the liquid refrigerant at high pressure suddenly transitions back to a low pressure, causing adiabatic flash evaporation of part of the liquid into a vapor. This reduces the temperature of the refrigerant, making it colder than e.g. the inside of the refrigerator and drawing in kinetic energy from the air inside said refrigerator before the vapor makes its way to the compressor again.
Elastocaloric Cooling
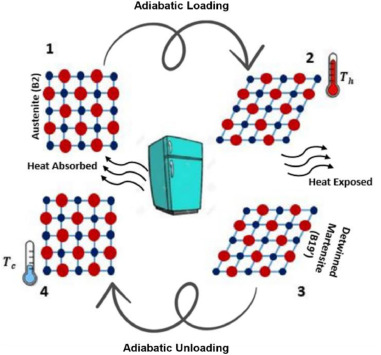
The elastocaloric effect. (Credit: Fatemeh Kordizadeh, Wikimedia)
With elastocaloric cooling (ECC) there is no liquid refrigerant or a pressure differential. Instead they rely on the elastocaloric effect, which is thermomechanical in nature.
Similar to how the refrigerant with vapor-compression refrigeration can absorb energy as it transitions from liquid to vapor and vice versa, with the elastocaloric effect it is the material itself that absorbs thermal energy from its environment when it’s mechanically loaded.
The aforementioned NiTi alloy is also known as a shape-memory alloy (SMA), which are generally known to be heat sensitive, finding use in applications like thermal fuses and sensors.
While the application of heat or cold can cause the deformation, this also works the other way around when mechanical force is applied. This is readily demonstrated with a strip of NiTi SMA and a thermal camera, as in this video by Helge Wurst:
As the strip is bent, the area experiencing the deformation becomes rather warm to the touch, with subsequent relaxation causing the same area to become cold to the touch.
Using such strips and mechanical actuators capable of applying 900 MPa of pressure, Guoan Zhou et al. were able to achieve freezing temperatures. They did this by combining multiple of such elastocaloric stages with CaCl2 as heat-exchange fluid. This is not a mainstream cooling method so far, but it should be quite reliable and low-maintenance.
Magnetocaloric Cooling
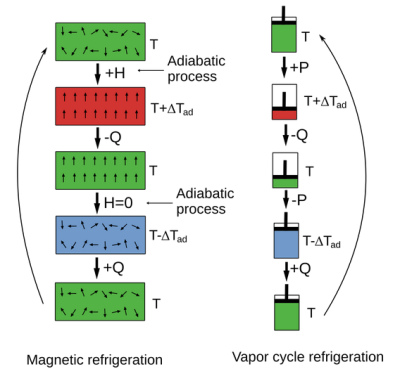
Comparison between magnetocaloric effect and vapor-compression cooling. (Source: Wikimedia)
The magnetocaloric effect (MCE) was first observed in 1881 by German physicist Emil Warburg, with the early 20th century seeing significant progress towards using it for cooling applications. This particular effect as the name suggests consists of exposing a material to a magnetic field, with this material then drawing in thermal energy. Upon removal of the magnetic field the material sheds this gained energy as well as some additional energy, thus cooling down relative to its environment.
Similar to the elastocaloric effect, this relies on an adiabatic process: without the transfer of any matter or entropy. This makes it a fully reversible process that can be repeated by successive applications of said magnetic field.
The biggest disadvantage with this effect for cooling purposes is that it’s only a very strong effect (giant MCE, or GMCE) in a limited number of alloys discovered so far. The first significant here was a rare-earth gadolinium-based alloy, Gd5(Si2Ge2), that showed GMCE at 270 K. This relatively low temperature and the use of rare-earths made this a tough sell.
More recently discovered alloys like Ni2Mn-X, where X is a variety of additives, display the GMCE near room temperature and even saw GE demonstrate an Ni-Mn-based magnetic refrigerator in 2014. So far commercialization of GMCE-based refrigeration is still rather limited but there is a push to make it work for generally less efficient vapor-compression-based home refrigerators.
Electrocaloric Cooling
Although easy to confuse with the magnetocaloric effect, the electrocaloric effect (ECE) pertains to the application of an electric field in dielectric materials. The effect is roughly the same, with the dipoles in the material either assuming an ordered or disordered state, depending on whether the field is respectively applied or turned off.
So far ECE-based cooling hasn’t seen commercialization yet either, though the past years there have been a range of breakthroughs, with for example Xin Chen et al. demonstrating ECE polymer films in 2023 that was subsequently used to create a thin-film refrigerator prototype with. This was claimed to achieve a Coefficient of Performance (COP) of a rather astounding 24, which compared to traditional heat pumps would make it a rather interesting solution if it can be commercialized.
Thermoelectric Cooling
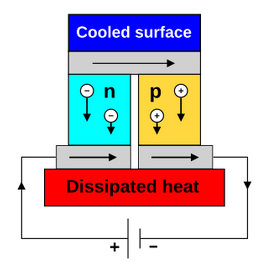
Diagram of a thermoelectric cooler. (Credit: Ken Brezier, Wikimedia)
The thermoelectric effect and the associated Peltier cooling devices are probably the most well-known and most heavily commercialized on this list along with vapor-compression. Within the thermoelectric effect, the Peltier effect concerns thermocouples and their associated temperature differences, thus lending its name to what are alternatively called ‘Peltier coolers’ as well as ‘thermoelectric coolers’, or TECs.
Rather than a refrigerant or rearranging of dipoles here the transfer of kinetic energy is performed using charge carriers within the TEC. On average charge carriers move to the ‘cool’ side, allowing them to transfer heat away from the other side.
As is well-known, this Peltier effect is rather limited when used as a heat pump, with very low efficiency and strict limitations on temperature differences. This is why their use in dehumidifiers and portable refrigerators is at best questionable.
The main reason why TECs are so popular can be said to be due to vapor-compression refrigeration being so bulky and neither elastocaloric, nor MCE, nor ECE solid-state coolers being quite ready for prime-time yet at the low-low price level that TECs can achieve due to being dead-simple semiconductor devices.
Pulse Tube Cooling
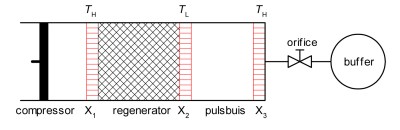
Stirling-type pulse tube refrigerator. (Credit: Mbeljaars, Wikimedia)
Another interesting, partially solid-state cooling method is the pulse tube refrigerator (PTR), which has seen limited use in commercial and other applications. Its main advantage is that it can be used as a cryocooler, making it ideal for space telescopes where sensors have to remain super-cold.
At its core it’s reminiscent of vapor-compression refrigerating, in that it uses a gas and a compressor, yet there’s no circulating loop of refrigerant. Inside the tube a piston alternately compresses the gas – often helium – which forces it through the regenerator. As the compression raises the temperature of the gas, this heat is then passed onto the material of the regenerator. On its way back through the regenerator this heat is then returned to the gas, explaining the name of this component.
The hot and cold sides of the regenerator are hereby used for cooling, though other PTR configurations are possible, such as the coaxial design. The relatively straightforward mechanical design and low temperatures achievable are why hobbyists are tinkering with PTRs in order to do things like making their own liquid nitrogen.
Chill Choices
Ultimately the question of what the right cooling method is for your particular task depends on a range of factors, including the required efficiency, available space and whether or not that big research grant budget just became available.
In terms of commercially available options that aren’t outrageously expensive, your options are somewhat limited, especially if you do not have a lot of space available. It’s possible that in a number of years these alternate technologies will be commercialized and wipe the floor with TECs in particular, but unless you’re currently heavily into tinkering with strips of NiTi SMA to build your own cooler, the primary options would seem to be either vapor-compression or TECs.
That said, considering that only a hundred years ago we were only just beginning to transition from iceboxes to vapor-compression refrigeration, it’s already pretty neat that we have some rather chill options to use today, and absolutely cool ones to look forward to.
Featured image: “Frosted Flakes“, National Park Service photo by [Neal Herbert]. Thumbnail image: “Frost” by [XoMEoX].
